New substance library to accelerate the search for active compounds
Substance library of the HZB-MX team is available worldwide for the development of drugs, also in the search for active substances against SARS-CoV-2
In order to accelerate the systematic development of drugs, the MX team at the Helmholtz-Zentrum Berlin (HZB) and the Drug Design Group at the University of Marburg have established a new substance library. It consists of 1103 organic molecules that could be used as building blocks for new drugs. The MX team has now validated this library in collaboration with the FragMAX group at MAX IV. The substance library of the HZB is available for research worldwide and also plays a role in the search for substances active against SARS-CoV-2.
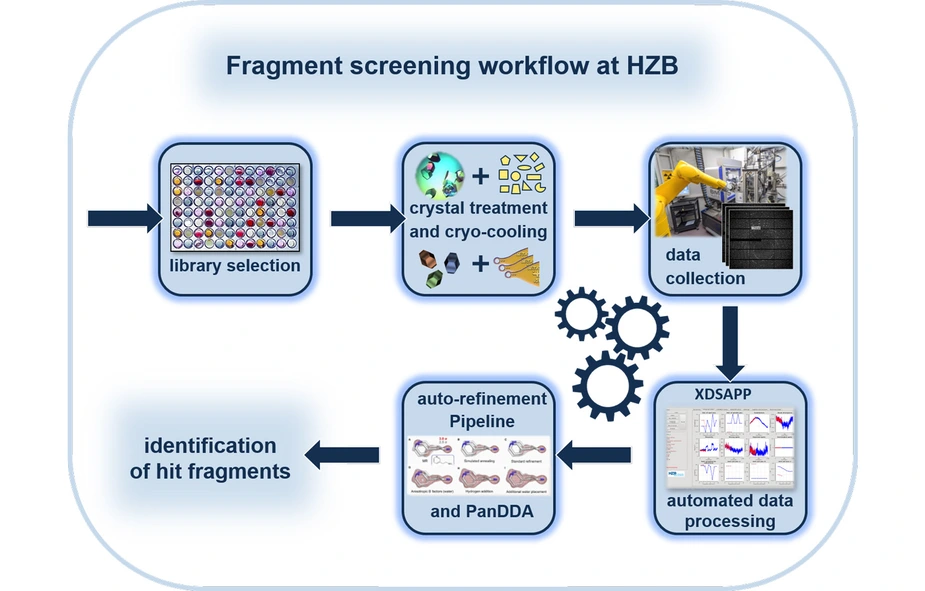
For drugs to be effective, they usually have to dock to proteins in the organism. Like a key in a lock, part of the drug molecule must fit into recesses or cavities of the target protein. For several years now, the team of the Macromolecular Crystallography Department (MX) at HZB headed by Dr. Manfred Weiss together with the Drug Design Group headed by Prof. Gerhard Klebe (University of Marburg) has therefore been working on building up what are known as fragment libraries. These consist of small organic molecules (fragments) with which the functionally important cavities on the surface of proteins can be probed and mapped. Protein crystals are saturated with the fragments and then analysed using powerful X-ray light. This allows three-dimensional structural information to be obtained at levels of atomic resolution. Among other things, it is possible to find out how well a specific molecule fragment docks to the target protein. The development of these substance libraries took place as part of the joint Frag4Lead research project and was funded by the German Federal Ministry of Education and Research (BMBF).
The MX team (MX stands for Macromolecular Crystallography) has now published the design of a chemically diverse fragment library called the “F2X-Universal“ library, which consists of 1,103 compounds. A representative selection of 96 compounds has been extracted from this library, which is referred to as the F2X Entry Screen. In the course of publishing the library, this selection has now been successfully tested and validated by the MX team of the HZB at the MAX IV X-ray source in Lund, Sweden and at BESSY II.
In the study, the HZB and MAX IV teams verified the efficiency of the F2X Entry library by screening endothiapepsin and the Aar2/RnaseH protein complex as the target enzymes. In the next step, the MX team will use the entire universal library.
“For the current study, the fragment screening experts at HZB - BESSY II worked very closely with the FragMAX project team at MAX IV“, said Dr. Uwe Müller from the MX team at HZB who helped to set up the three MX beamlines at BESSY II as well as the BioMAX beamline at MAX IV. “This enabled both partners to further develop their own technology platforms and use them for imaging the functional surfaces of different proteins. This will be an excellent basis for future collaboration between MAX IV and HZB.“
Publication:
Structure (2020): F2X-Universal and F2X-Entry: Structurally Diverse Compound Libraries for Crystallographic Fragment Screening. Jan Wollenhaupt, Alexander Metz, Tatjana Barthel, Gustavo Lima, Andreas Heine, Uwe Mueller, Gerhard Klebe, Manfred S. Weiss
DOI: 10.1016/j.str.2020.04.019
Contact:
Helmholtz-Zentrum Berlin für Materialien und Energie
Research Group Macromolecular Crystallography
Dr. Jan Wollenhaupt
Phone: +49 30 8062-13483
Email: jan.wollenhaupt(at)helmholtz-berlin.de
Dr. Manfred Weiss
Phone: +49 30 8062-13149
Email: manfred.weiss(at)helmholtz-berlin.de